The Real Root of Chronic Disease: It’s Not What You Think
For decades, we’ve labeled conditions like obesity, diabetes, cardiovascular disease, and dementia as separate “diseases.” Entire industries have been built around managing them individually—cardiology for the heart, endocrinology for metabolism, neurology for the brain.
But what if that framework is fundamentally incomplete?
What if these aren’t distinct diseases at all—but different expressions of the same underlying cellular dysfunction?
This is the paradigm shift advanced by Dr. Robert Lustig, and articulated in Metabolical, particularly in Chapter 7: “The Diseases that Aren’t Diseases.” In that chapter, Lustig identifies eight core intracellular processes that underpin most chronic disease. When these processes are disrupted, pathology emerges—regardless of which organ system ultimately breaks down.
One Unifying Mechanism: Cellular Energy Breakdown
At the center of this model is the mitochondrion—the engine of the cell.
Healthy mitochondria generate ATP (cellular energy), regulate signaling, and maintain metabolic balance. But when they are overwhelmed or impaired, the entire system begins to destabilize.
One of the most important drivers of this breakdown is:
Reactive Oxygen Species (ROS).
ROS are not inherently harmful. In fact, at low levels they serve as essential signaling molecules. But when produced in excess, they overwhelm the system—damaging proteins, lipids, DNA, and the mitochondria themselves.
This is where the story of chronic disease begins to converge.
ROS: The Common Pathway
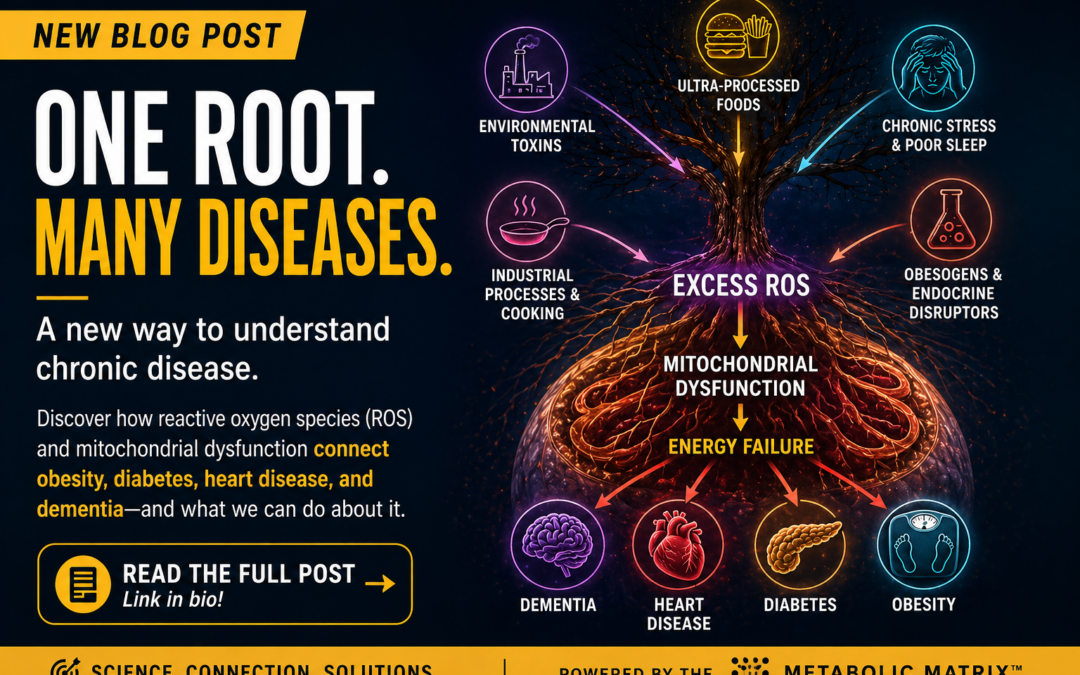
The infographic accompanying this piece highlights a critical insight:
A wide range of modern exposures all converge on one shared pathway—excess ROS generation.
From the research presented:
- Environmental toxins and “obesogens” disrupt hormonal and metabolic signaling while increasing oxidative stress
- Ultra-processed foods—especially those high in fructose—impair mitochondrial function and elevate ROS
- Chronic stress and sleep deprivation increase energy demand while impairing recovery and repair
- Industrial processing and high-heat cooking generate compounds that further drive oxidative load
These factors don’t operate in isolation—they accumulate.
And they all point in the same direction: mitochondrial dysfunction and metabolic imbalance.
From Energy Failure to Disease
When ROS exceed the body’s capacity to manage them, two critical failures occur:
- ATP production declines — cells can’t generate sufficient energy
- ATP demand increases — due to stress, inflammation, and repair processes
This mismatch is particularly damaging in energy-intensive tissues like the brain.
As outlined in Dr. Lustig’s model:
- Reduced ATP compromises neuronal function
- Excess ROS activates inflammatory pathways (e.g., NF-κB)
- Protein misfolding and plaque formation accelerate
- Neurons ultimately fail and die
In this context, dementia is not an isolated neurological condition—it is the downstream consequence of systemic metabolic dysfunction.
Reframing Chronic Disease
This brings us back to Metabolical.
In Chapter 7, Lustig reframes chronic disease as a breakdown across eight key intracellular processes, including:
- Energy production
- Oxidative stress balance
- Inflammation signaling
- Hormonal regulation
- Nutrient sensing
When these processes are disrupted, different tissues manifest disease in different ways—but the root cause remains shared.
So instead of asking:
“What disease does this patient have?”
We should be asking:
“Which cellular processes are being disrupted—and what is driving that disruption?”
Why This Matters
This reframing has profound implications.
It means:
- Obesity is not simply about excess calories
- Diabetes is not just about glucose
- Dementia is not just about the brain
They are all manifestations of system-wide metabolic dysfunction, driven by the cumulative impact of modern diet, environment, and lifestyle.
And importantly, it means intervention is possible—not just downstream, but at the source.
Because if the root is cellular, then the solution must also be systemic:
- Reduce inputs that drive excess ROS
- Restore mitochondrial function
- Support the body’s natural repair and regulatory systems
From Insight to Action
Understanding the science is only the beginning.
If chronic disease is driven by disruptions in core cellular processes—energy production, oxidative balance, inflammation, and signaling—then the critical question becomes:
How do we translate this into action at scale?
This is where the Metabolic Matrix comes in.
The Metabolic Matrix converts this complex biology—grounded in Dr. Lustig’s work and detailed in Metabolical—into a practical, systems-level framework. It maps how ingredients, processing methods, and product design influence the intracellular pathways that determine health outcomes.
It bridges the gap between:
- Mechanism (ROS, mitochondrial function, metabolic signaling)
and - Execution (what gets formulated, produced, and consumed)
For the food and beverage industry, this represents a fundamental shift.
It moves beyond:
- calories
- macronutrients
- “better-for-you” claims
…and toward a more rigorous standard:
Does this product support or disrupt human cellular function?
Because the same drivers of dysfunction—oxidative stress, mitochondrial impairment, inflammatory signaling—are shaped upstream by formulation choices, sourcing decisions, and processing techniques.
The Metabolic Matrix makes this actionable.
It provides a framework that can be applied across any portfolio, enabling companies to:
- identify hidden metabolic disruptors
- reformulate with biological intent
- design products aligned with human physiology
A New Standard for Health
If the science is clear—and increasingly, it is—then the opportunity is equally clear.
We can continue managing chronic disease after it emerges.
Or we can redesign the upstream inputs that drive it.
The Metabolic Matrix offers a pathway to do exactly that—translating metabolic science into practical decisions that the food and beverage industry can implement today, across any portfolio.
Because ultimately, the future of health won’t be built in the clinic.
It will be built in the systems that determine what—and how—we eat.