There are moments in science when the problem is not a lack of data, but a failure of framing.
Nutrition science appears to be living through one of those moments now.
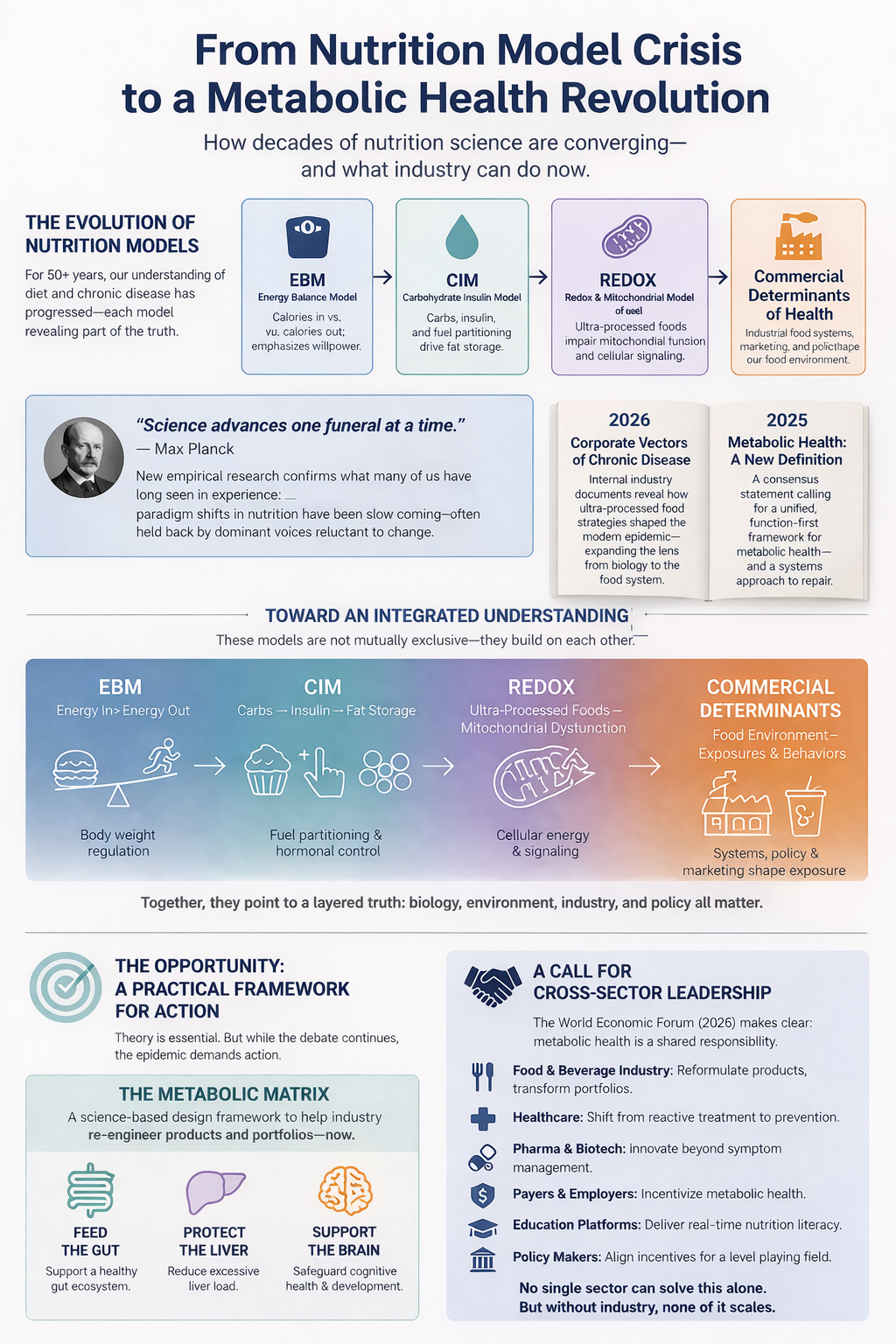
For decades, the field has argued over obesity and metabolic disease through increasingly polarized mechanistic models. The Energy Balance Model (EBM) has long held institutional dominance, locating the central cause of obesity in chronic positive energy balance, with weight gain driven by overeating relative to expenditure. The Carbohydrate-Insulin Model (CIM) emerged as a major challenge, arguing that diet composition—especially high-glycemic-load foods—drives hormonal responses that shift fuel partitioning toward fat storage, with positive energy balance arising downstream rather than upstream. More recently, the REDOX model and the broader obesogen framework have expanded the debate further, bringing mitochondrial signaling, oxidative stress, endocrine disruption, developmental programming, and environmental chemical exposure into the causal architecture.
A new Cell Metabolism forum by David Ludwig, Overcoming Impasse in Nutrition Science, gives this historical moment an unusually precise name: impasse. Ludwig argues that paradigm clash is a necessary and even salutary part of science, but that in nutrition research it has too often hardened into inconclusive conflict, with debates that persist for decades without clear resolution, to the detriment of both scientific progress and public health. Using obesity as a case study, he explicitly frames the field through Thomas Kuhn’s model of scientific change: pre-paradigm description, normal science, model drift, crisis, model revolution, and sometimes paradigm change.
That framing is hard to ignore because it captures something many scientists, clinicians, strategists, and public health thinkers have sensed for years: obesity science has not merely been debating evidence; it has been debating causality from within rival conceptual worlds.
The Long Reign of Normal Science
Ludwig’s essay is especially useful because it does not begin with team sports. It begins with the structure of science itself.
He notes that much obesity thinking remains partially descriptive rather than causally anchored. Statements such as “obesity pathogenesis involves sustained positive energy balance” or that behavioral changes creating an energy deficit produce weight loss are, in his reading, conceptually incomplete because they describe a physical bookkeeping relationship rather than specify directional causality. The law of energy conservation tells us that changes in stored body energy and energy balance must be equal; it does not tell us what initiates the process. To create a causal model, a directional arrow must be supplied by biology, not by physics alone.
That distinction matters.
The EBM, in its dominant form, supplies the arrow this way: people gain weight because they consume more energy than they expend, in a food environment rich in convenient, highly palatable, energy-dense products. In that model, excess intake is primary, fat storage is downstream, and the practical response is some variation on “eat less, move more.” Ludwig describes this as the prevailing normal-science paradigm.
Hall and colleagues, defending a more sophisticated contemporary version of the EBM, object strongly to the caricature that EBM is merely a slogan about willpower. In their 2022 paper, they argue that the EBM is a biologically grounded model in which the brain is the primary organ regulating body weight, integrating internal and external signals, largely below conscious awareness, to control food intake over time. They also argue that the modern food environment—especially the widespread availability and marketing of inexpensive, convenient, energy-dense, ultra-processed foods high in fat and sugar and low in protein and fiber—is a likely major driver of the rise in obesity prevalence.
That is an important clarification. It means the serious form of the EBM is not reducible to moralizing slogans or simplistic calorie arithmetic. It already includes the brain, endocrine signaling, food reward, food environment, and individual susceptibility.
And yet, Ludwig’s point remains: even refined versions of the dominant model have struggled to resolve persistent anomalies. Why did decades of energy-restriction-centered obesity treatment produce such poor long-term outcomes? Why does the body so vigorously defend a higher weight once achieved? Why are prevention efforts so difficult if the average annual gain can arise from what appears to be such a small daily energy excess? Why do animals and humans frequently return toward a defended baseline after overfeeding experiments? And why has the focus on calorie restriction not come close to stemming the epidemic?
These are not trivial loose ends. In Kuhnian terms, they are anomalies.
The Carbohydrate-Insulin Revolt
The CIM emerged to reverse the arrow.
In Ludwig’s formulation, a high-glycemic-load meal shifts postprandial hormonal signaling—especially the insulin-to-glucagon balance—toward storage rather than oxidation. As a result, more fuel is driven into adipose tissue, leaving less readily available for oxidation in other tissues. Hunger then rises, food reward and salience increase, or energy expenditure falls in compensation. In this model, fat deposition drives positive energy balance, not the other way around. Ludwig notes that this causal direction has analogies elsewhere in physiology and pathology, including adolescence, pregnancy, and tissue growth states in which energy deposition into new tissue drives increased intake.
Whether one agrees with every detail or not, the CIM performed an essential function in the field. It forced serious attention to diet quality, glycemic load, substrate partitioning, insulin dynamics, and the endocrine consequences of ultra-processed carbohydrate-rich dietary patterns. It challenged the assumption that calories are practically interchangeable for body-weight regulation. It reopened the question of whether obesity is driven only by excess intake or whether internal metabolic partitioning can help drive intake itself.
Hall and colleagues push back hard. In their critique, all obesity models must satisfy energy conservation, and the updated CIM has effectively abandoned some of its earlier defining features, becoming in their view a narrower special case within a broader EBM framework focused mainly on glycemic load. They further argue that data from animal studies, population trends, and intervention trials do not support the claim that carbohydrate intake is the primary driver of the obesity epidemic. They point to evidence that long-term weight change correlates with multiple aspects of diet quality and food patterning, not simply carbohydrate quantity, and that low-glycemic-load diets do not generally produce greater long-term weight loss than higher-glycemic-load diets under controlled trial conditions.
This is precisely the kind of exchange Ludwig identifies as prone to paralysis. Rival camps may share datasets, methods, and terminology, yet still remain partly at cross-purposes because they are weighing anomalies differently, emphasizing different endpoints, and arguing from within partly incompatible conceptual assumptions. Ludwig explicitly invokes Kuhn’s concept of incommensurability to explain why such arguments so often fail to converge. Competing paradigms, he notes, frequently disagree not only on answers but on what counts as the relevant problem to be solved, what standards of evidence matter most, and how apparently shared terms are to be interpreted.
The Sociology of Scientific Delay
At this point, Max Planck enters the room.
Planck’s famous observation—“science advances one funeral at a time”—has often been quoted as a cynical joke about intellectual stubbornness. But the Azoulay, Fons-Rosen, and Graff Zivin paper, Does Science Advance One Funeral at a Time?, gives it empirical weight. Studying the premature death of eminent life scientists, the authors found that publication by collaborators in affected fields fell, but publication by outsiders rose markedly. Those outsider contributions drew more heavily on different scientific corpora and were disproportionately likely to be highly cited. The authors conclude that the loss of a luminary can create space for fields to evolve in new directions that advance the frontier.
Notably, the mechanism does not seem to be primarily direct gatekeeping. The paper suggests that the very presence of a dominant figure may deter outsiders from entering a field at all. The towering scholar’s shadow alters the cost-benefit calculus of challenge. Outsiders wait, redirect, or stay away. When that shadow lifts, the field becomes more permeable.
This is relevant because the obesity field has not simply been a technical dispute. It has also been a social one. Prestige, institutional alignment, editorial influence, intellectual camps, and reputational risk have all shaped the tempo of change. Ludwig’s essay offers a field-specific diagnosis of impasse; the Azoulay paper offers a broader sociology of why impasse can persist even when anomaly accumulation has become obvious.
Why Neither EBM nor CIM Is Sufficient
And yet, even if one grants that CIM was historically necessary, it does not solve everything.
This is where the field becomes more interesting—and more promising.
The Heindel, Lustig, Howard, and Corkey paper, Obesogens: a unifying theory for the global rise in obesity, argues that the obesity pandemic is unlikely to be explained adequately by either EBM or CIM alone. The paper proposes a combined OBS/REDOX model linking four frameworks: EBM, CIM, REDOX, and the obesogen model. In that synthesis, environmental chemicals present in air, food, food packaging, and household products generate false autocrine and endocrine signals, including reactive oxygen species (ROS), that distort standard regulatory energy mechanisms, increase insulin secretion, disrupt energy efficiency, alter appetite and expenditure, and thereby contribute to adiposity. The authors explicitly state that this combined model incorporates data supporting both EBM and CIM while extending the field into environmental and developmental causation.
This matters for several reasons.
First, the paper places developmental programming at the center of the obesity conversation. It argues that obesity can begin in utero and early life, with nutrition, stress, and environmental chemicals altering epigenetic regulation, adipose differentiation, metabolic tissue development, and long-term susceptibility to weight gain across the lifespan and even across generations.
Second, it offers a biochemical bridge through REDOX signaling. Excess fuel, or chemicals that mimic false energy-surplus signaling, can increase ROS and alter redox balance. In this model, ROS is not only a marker of damage but also a signaling event that affects insulin secretion, fat storage, satiety pathways, and metabolic efficiency. The paper notes that pancreatic beta cells have limited ROS-scavenging capacity, making insulin secretion an especially early and sensitive response to excess ROS from nutrients or obesogens. It also states that ROS can directly stimulate insulin secretion even in the absence of glucose and that removing ROS can reduce insulin secretion.
Third, the model helps explain phenomena that neither EBM nor CIM fully captures alone. It provides a rationale for why obesogens might influence adipocyte number, size, location, and function; food intake; energy expenditure; insulin resistance; inflammation; microbiome alteration; and long-term set-point vulnerability. The authors explicitly note that obesogens may help explain why animals sharing human environments, including animals in captivity with controlled diets, have also gained weight over recent decades.
Fourth, it directly implicates the ultra-processed food environment in more than one way. The Western diet is described as obesogenic not only through nutrient structure but also through the presence of chemicals in food packaging, additives, preservatives, emulsifiers, antioxidants, and pesticide residues. Ultra-processed food becomes not only a dietary object but also a chemical delivery system.
This is a major advance because it shifts the conversation from model rivalry to layered causation.
Dr. Robert H. Lustig’s master work “Metabolical: The Lure and the Lies of Processed Food, Nutrition, and Modern Medicine” is essential reading, and in Chapter 7, Dr. Lustig argues that chronic disease stems from eight specific subcellular pathologies—glycation, oxidative stress, mitochondrial dysfunction, insulin resistance, membrane instability, inflammation, epigenetic changes, and impaired autophagy—driven by ultra-processed food consumption. The chapter emphasizes that these pathologies, which collectively represent cellular dysfunction, can be mitigated by a “feed the gut, protect the liver” approach focused on high-fiber, low-sugar, and real food diets.
From Internal Biology to External Exposure
But even OBS/REDOX, powerful as it is, remains largely a model of internal pathophysiology. It explains how bodies may be mis-signaled, injured, programmed, and pushed toward adiposity. It does not by itself fully explain the system producing the exposures in the first place.
That is why the new New England Journal of Medicine paper, “Corporate Vectors of Chronic Disease — Using Internal Industry Documents to Craft Counterstrategies,” is so important.
This paper broadens the frame from obesity mechanism to disease vector. It argues that transnational corporations that manufacture and market health-harming products function as major vectors of chronic disease. Chronic diseases, including cancers, diabetes, neurocognitive disorders, and chronic respiratory disease, account for the large majority of global deaths, and the paper identifies five commercial product categories—fossil fuels, tobacco, ultra-processed foods, chemicals, and alcohol—as key contributors to this burden, alongside drug overdose. The authors place these harms within the now-familiar language of the commercial determinants of health.
Most importantly, the paper identifies three common mechanisms used across major health-harming industries:
- Knowledge capture
- Regulatory or policy capture
- Shaping of the public narrative
These mechanisms are not abstract.
Under knowledge capture, the paper describes how corporations influence research agendas, suppress unfavorable findings, attack independent scientists, and sponsor studies that downplay harm. One example is the internal chemical-industry knowledge surrounding PFAS toxicity: DuPont and 3M had evidence of harm decades before public action, yet suppressed and obscured the data while widespread exposure continued.
Under narrative shaping, the paper shows how tactics migrate across industries. Tobacco companies not only sold cigarettes; they also acquired and reformulated food and beverage products, including children’s sugary drinks, then leveraged tobacco-style marketing and product engineering to expand exposure and increase appeal. The examples of Hawaiian Punch and Kool-Aid line extensions make plain that the industrial playbook is portable.
Under regulatory capture, the paper links commercial determinants to political determinants, showing how lobbying, trade associations, revolving doors, and funding structures help shape regulatory environments that preserve harmful product markets. It also recommends counterstrategies including public transparency around industry funding, mandatory data sharing and study registration, and structural separation between industry money and health research through independent public mechanisms.
This is where the overall story sharpens.
The debates among EBM, CIM, REDOX, and OBS are largely debates about what happens after exposure.
The NEJM paper is about who manufactures, scales, protects, and normalizes the exposures.
That is the missing outer ring.
The Fire, the Wiring, and the Arsonist
One could say that the obesity field has spent decades arguing over the wiring diagram while the building burns.
Did excess calories start the fire?
Did insulin start it?
Did mitochondrial signaling start it?
Did endocrine-disrupting chemicals start it?
These are all serious questions. They remain worth debating. Ludwig is right that paradigm clash can be constructive and that the field should not fear foundational argument.
But the NEJM paper forces an additional question:
Who keeps pouring accelerant into the building?
Once the outer systems layer is included, the scientific puzzle begins to look different. The epidemic no longer appears to be only a question of internal mechanism. It appears to be a stacked system of exposures and incentives:
- industrial food formulation
- chemical contamination and packaging
- ultra-processed dietary patterning
- developmental and intergenerational programming
- commercial and regulatory capture
- narrative distortion
- vulnerable populations living inside exposure-heavy environments
The result is that bodies do not merely “choose badly” in an otherwise neutral world. They are immersed in a world designed to produce metabolic risk.
Why an Integrative Framework Is Now Needed
If this reading is correct, then the next step in obesity science is probably not the final triumph of one model over all others.
It is probably the emergence of an integrative framework capable of situating each model within a larger causal architecture.
Such a framework would recognize that:
The EBM captures important truths about energy regulation, food environment, food reward, and the central role of neural integration.
The CIM captures important truths about glycemic load, hormonal response, fuel partitioning, and the possibility that fat storage may help drive intake and expenditure adaptation.
The REDOX model captures important truths about mitochondrial signaling, ROS, false energy-state information, and metabolic dysregulation.
The obesogen model captures important truths about endocrine disruption, chemical exposures, developmental programming, epigenetics, and altered lifelong susceptibility to weight gain.
The commercial determinants of health framework captures important truths about the industrial production, political protection, and public normalization of health-harming exposures.
Put differently: the field may be nearing the point where the real advance is no longer choosing among rival internal models, but placing them in relation to each other and to the external systems that generate the burden of exposure.
Why Action Cannot Wait for Total Consensus
This has practical implications.
One recurrent argument within food and beverage circles has been that metabolic health should not yet be directly addressed because there is insufficient consensus: not enough consensus on the dominant obesity model, not enough consensus on the exact biomarkers, not enough consensus on how to define metabolic health operationally across populations.
That objection is becoming less persuasive.
The reason is simple: waiting for perfect theoretical consensus is not the same thing as requiring sufficient evidence for practical harm reduction. The history of public health is full of cases where action preceded total mechanistic closure. The NEJM paper itself points to tobacco as the instructive example: research on internal industry documents, coupled with policy action, changed the terrain long before every scientific dispute had been neatly settled.
In metabolic health, the fire is already visible.
There is already strong reason to reduce the burden of ultra-processed foods, improve food structure and quality, lower unnecessary glycemic insult where relevant, eliminate or reduce known and suspected harmful additives, reduce exposure to endocrine-disrupting and mitochondria-disrupting chemicals, improve fiber, protein, and micronutrient density, and stop allowing commercial capture to masquerade as uncertainty. Those interventions do not require the field to stop debating. They require the field—and especially industry—to stop hiding behind debate.
From Model War to Model Architecture
This is the deeper significance of the present moment.
Ludwig’s essay names the impasse.
Azoulay gives empirical support to Planck’s suspicion that fields may evolve only when intellectual space opens.
Heindel, Lustig, Howard, and Corkey offer a serious effort at mechanistic synthesis.
The NEJM paper expands the frame outward to the corporate production of exposure itself.
Taken together, these works suggest that the next paradigm in obesity and metabolic disease may not be a single-cause theory at all.
It may be a layered systems model capable of holding:
- developmental biology
- endocrinology
- mitochondrial signaling
- toxicology
- diet structure
- food processing
- commercial determinants
- regulatory capture
- public health intervention
- and product redesign
within one coherent causal map.
That would be a genuine scientific advance.
Not because it would end debate, but because it would place debate within a larger architecture that is truer to the complexity of the problem.
The building is on fire. It remains useful to understand whether the wiring, the fuel source, the insulation, or the airflow explains the flames. But it is no less necessary to identify who designed the building materials, who sold the accelerants, who lobbied against sprinklers, who funded the misinformation, and who profited while the alarms were muted.
That is where metabolic science now stands.
The future will belong not to the loudest model, but to the framework capable of integrating biological mechanism with environmental exposure, industrial causation, and practical redesign.
And that future cannot wait for the next funeral.
The recent World Economic Forum briefing, Catalysing Cross-Sector Leadership for Metabolic Health, makes the case that metabolic health is not a niche clinical issue but a defining societal and economic challenge. It frames metabolic health as the body’s ability to efficiently produce, regulate, and use energy across the life course, links dysfunction to risks across cardiovascular, renal, hepatic, glycaemic, and inflammatory pathways, and argues that neither treatment alone nor GLP-1 therapies alone will be sufficient at population scale. Instead, the brief calls for a holistic, multi-sector approach in which food manufacturers, healthcare providers, pharmaceutical companies, insurers, consumer-health platforms, and public institutions each play distinct but interconnected roles. For industry specifically, it highlights the need for healthier product reformulation, portfolio transformation, institutional nutrition partnerships, real-time nutrition education, shared metrics, and collaborative action at scale, all within a level playing field so that no company is penalized for moving first.
This is precisely where the Metabolic Matrix becomes so valuable. As important as EBM, CIM, REDOX, obesogen science, and the commercial determinants framework all are, the Metabolic Matrix was built not to compete with them, but to operationalize their implications for industry now. The Matrix starts from three practical scientific pillars—feed the gut, protect the liver, support the brain—and translates them into criteria, metrics, tiering, ingredient review, product evaluation, and reformulation pathways that companies can actually use across portfolios. It does not conflict with the other models; it builds on them. If EBM helps explain energy regulation, CIM fuel partitioning, REDOX mitochondrial signaling, and obesogen science developmental and environmental disruption, then the Metabolic Matrix is the translational design framework that turns those insights into product reformulation, portfolio redesign, and measurable commercial action. In that sense, it is not another theory in the model wars. It is a tool for ending paralysis and helping industry begin the work of metabolic repair now.
References
Azoulay, Pierre, Christian Fons-Rosen, and Joshua S. Graff Zivin. “Does Science Advance One Funeral at a Time?” American Economic Review 109, no. 8 (2019): 2889–2920. This paper examines the premature death of eminent life scientists and finds that outsider contributions into affected subfields rise after their passing, often drawing on different scientific corpora and receiving high citation impact.
Hall, Kevin D., I. Sadaf Farooqi, Jeffery M. Friedman, Samuel Klein, Ruth J.F. Loos, David J. Mangelsdorf, Stephen O’Rahilly, Eric Ravussin, Leanne M. Redman, Donna H. Ryan, John R. Speakman, and Deirdre K. Tobias. “The Energy Balance Model of Obesity: Beyond Calories In, Calories Out.” American Journal of Clinical Nutrition 115 (2022): 1243–1254. This paper presents a biologically grounded contemporary version of the EBM, emphasizing brain-mediated regulation of body weight and the role of the modern food environment. It also argues that the EBM is more robust than the CIM.
Metabolic Matrix methods paper
Harlan TS, Gow RV, Kornstädt A, Alderson PW, Lustig RH. The Metabolic Matrix: Re-engineering ultraprocessed foods to feed the gut, protect the liver, and support the brain. Frontiers in Nutrition. 2023;10:1098453. doi:10.3389/fnut.2023.1098453. Available at: Frontiers in Nutrition – The Metabolic Matrix.
Heindel, Jerrold J., Robert H. Lustig, Sarah Howard, and Barbara E. Corkey. “Obesogens: A Unifying Theory for the Global Rise in Obesity.” International Journal of Obesity 48 (2024): 449–460. This review proposes the combined OBS/REDOX model integrating EBM, CIM, REDOX, and obesogen science, with particular emphasis on developmental programming, environmental chemicals, ROS signaling, and mitochondrial dysfunction.
Khettab, Sid Ahmed. “The Evolution of Science: The Kuhn Cycle.” Clevious Discourse (2020). This secondary explanatory piece provides a simplified presentation of the Kuhn cycle—normal science, model drift, crisis, revolution, and paradigm shift—which is useful as a pedagogic shorthand, though Kuhn’s original work remains the more authoritative source.
Kuhn, Thomas S. The Structure of Scientific Revolutions. 2nd ed. Chicago: University of Chicago Press, 1970. Kuhn’s ideas are discussed extensively in Ludwig’s 2026 essay, especially the concepts of normal science, anomaly accumulation, crisis, revolution, and incommensurability.
Ludwig, David S. “Overcoming Impasse in Nutrition Science.” Cell Metabolism 38 (2026): 452–455. Ludwig applies Kuhn’s framework directly to obesity science, diagnosing prolonged paradigm clash between the EBM and CIM and calling for the field to develop better ways of resolving disputes and overcoming stalemate.
Consortium of the Center to End Corporate Harm, University of California, San Francisco. “Corporate Vectors of Chronic Disease — Using Internal Industry Documents to Craft Counterstrategies.” New England Journal of Medicine 394, no. 12 (2026): 1231–1236. This paper argues that transnational corporations producing health-harming products act as major vectors of chronic disease and identifies shared tactics across industries, including knowledge capture, policy capture, and narrative shaping. It also proposes counterstrategies to reduce corporate influence on health research and policy.
World Economic Forum / McKinsey Health Institute brief
World Economic Forum. Catalysing Cross-Sector Leadership for Metabolic Health. Geneva: World Economic Forum; 2026. Available at: WEF Catalysing Cross-Sector Leadership for Metabolic Health (PDF). Accessed March 26, 2026.
